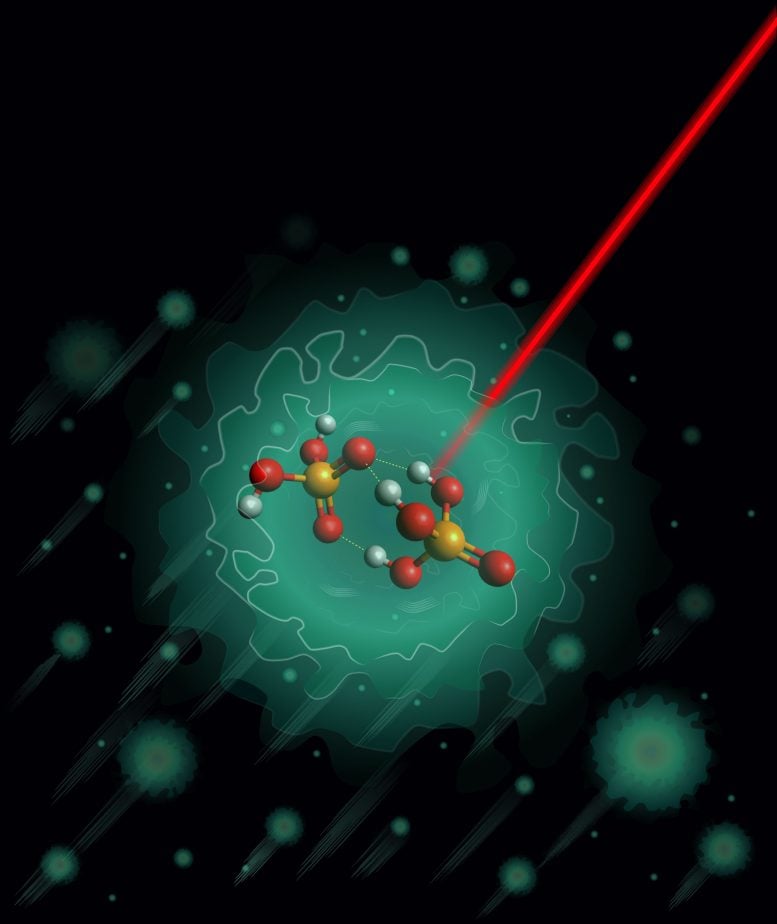
By freezing a crucial phosphoric acid complex to near absolute zero, scientists uncovered a single, unexpectedly stable structure at the heart of proton transport.
Phosphoric acid is vital in both biology and modern technology because of its exceptional ability to move electrical charge. Inside the human body and in devices such as fuel cells, this small molecule helps drive essential chemical reactions.
Scientists at the Department of Molecular Physics at the Fritz Haber Institute have now uncovered new details about how it performs this task at the molecular level.
How Tiny Electrical Signals Control Life
Every second, countless electrical charges flow through our bodies. These signals are essential for life. Processes such as cellular communication, energy conversion, and metabolism all rely on the carefully controlled movement of charged particles across membranes and within cells. In many ways, the transport of charge serves as a fundamental regulatory system.
Phosphoric acid (H3PO4) and related phosphate compounds are found throughout living organisms. They form the backbone of DNA and RNA, contribute to the structure of cell membranes, and are part of ATP, the molecule that stores and delivers energy in cells. These compounds are especially important for moving positive charges in biological systems.
Beyond biology, phosphoric acid also plays a significant technological role. It is used in certain types of batteries and in fuel cells. In these systems, engineers take advantage of one standout feature: its unusually high proton conductivity.
Protons carry a positive charge and can move through phosphate-containing materials in a stepwise fashion. They “jump” from one molecule to the next along networks of hydrogen bonds. This process, known as “proton-shuttling”, enables charges to travel extremely quickly.
Although scientists have long understood that this mechanism operates, many of its finer details have remained unclear. In their recent study, researchers from the Fritz Haber Institute, working with collaborators in Leipzig and the USA, focused on determining the structure of a crucial negatively charged phosphoric acid complex. By clarifying its structure, they aimed to better understand the earliest stages of proton transfer.
A Cold Look at Hot Chemistry with Cryogenic Spectroscopy
Earlier research suggested that a specific negatively charged form of phosphoric acid could act as the starting point for the proton-shuttling sequence. This species is the deprotonated dimer H3PO4·H2PO4-.
To examine it more closely, the team created this molecule in the laboratory and studied it under extremely cold conditions. They embedded it inside a helium nanodroplet, cooling it to just 0.37 degrees above absolute zero. Using infrared radiation, they then analyzed its structure.
Cooling the molecule to such low temperatures greatly reduces thermal motion and other disturbances. This allows scientists to measure its structure with high precision. The experimental findings were further supported by quantum chemical calculations, which help predict how molecules are arranged and how they behave.
The Invisible Network: Structure and Hydrogen Bonds Found
When the researchers compared their measurements with theoretical predictions, they found only partial agreement. Computational models had suggested that two structural forms should be equally likely. However, the experimental results clearly showed that the deprotonated phosphoric acid dimer adopts a single stable structure.
This structure is relatively rigid and presents high energy barriers for proton transfer. It contains three hydrogen bonds and features a shared oxygen atom that serves as an acceptor. Similar arrangements have been observed in other phosphoric acid-containing clusters, indicating that this hydrogen-bonding pattern may be common in such systems.
The findings demonstrate that theoretical models alone may not always capture the full picture. Careful experimental work remains essential for determining accurate molecular structures.
Why It Matters
The study offers new insight into the molecular basis of phosphoric acid’s remarkable proton conductivity, “Nature’s proton highway.” By identifying a single stable structure for the key anionic dimer H3PO4·H2PO4- and revealing its distinct hydrogen-bonding motif, the researchers have clarified an important piece of the proton transport puzzle.
In addition, the work provides a valuable reference point for improving quantum chemical methods used to model phosphate-containing clusters. These advances could support the development of better proton-conducting materials and deepen scientific understanding of how proton transfer operates in living systems.
Reference: “Cryogenic Vibrational Spectroscopy of the Deprotonated Dimer of Phosphoric Acid” by América Y. Torres-Boy, Jia Han, Gurpur Rakesh D. Prabhu, Martín I. Taccone, Anoushka Ghosh, Hannah Buttkus, Katja Ober, Gerard Meijer, Knut R. Asmis, Anne B. McCoy and Gert von Helden, 15 December 2025, The Journal of Physical Chemistry A.
DOI: 10.1021/acs.jpca.5c06704
Funding: Deutsche Forschungsgemeinschaft, Alexander von Humboldt Foundation, DAAD-RISE funding
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.
