What do plants, toads, and mushrooms have in common? They can all produce psychedelic substances – and now their powers have been combined in one plant, like a trippier Captain Planet.
In a wild first, scientists have taken the genes these organisms use to make five natural psychedelics and introduced them into a tobacco plant (Nicotiana benthamiana), which then produced all five compounds simultaneously.
As interest grows in psychedelics as potential treatments for illnesses such as depression, anxiety, and PTSD, the newly developed system could offer scientists a new way to produce these compounds for research purposes.
“[Our] strategy established a heterologous plant system for the production of five prominent therapeutically valuable compounds, their derivatives, and nonnatural plant analogs, providing a starting point for their production in plants,” writes a team led by researchers at the Weizmann Institute of Science in Israel.
Tryptamine psychedelics are a class of compounds that includes psilocin, psilocybin, and a number of dimethyltryptamine (DMT) compounds. The ability to produce these substances has emerged in diverse organisms across the tree of life – plants, fungi, and animals.
In recent years, a number of studies have shown that tryptamine psychedelics may represent an untapped resource when it comes to mental health treatments.
However, progress in this field remains limited, in part due to regulatory restrictions, underscoring the need for more research. This creates practical challenges for scientists.
“Traditionally, the supply of psychedelics relies on natural producers, mainly plants, fungi, and the Sonoran Desert toad,” the researchers write.
“Harvesting these organisms for their psychoactive compounds raises ecological and ethical concerns, being increasingly threatened by habitat loss and overexploitation.”
In an effort to tackle this, plant scientists Paula Berman and Janka Höfer and their team set out to map and rebuild the biochemical pathways behind these compounds.
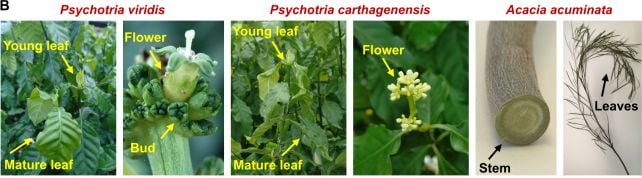
They identified the key genes used by two plants – Psychotria viridis and Acacia acuminata – to make DMT, and the step-by-step chemical pathways involved in producing the compound.
Then, they combined these with genes and pathways already known from psychedelic mushrooms (Psilocybe cubensis) and the cane toad (Rhinella marina), added supporting enzymes from rice and cress, then genetically introduced the combined genetic toolkit and kaboodle into tobacco plants (Nicotiana benthamiana).
The tobacco was chosen not because of its own drug production, but because it’s basically the lab rat of plant species, with its fast growth.
Finally, the team carefully monitored the plant’s production of five psychedelic tryptamines: DMT originally from plants; psilocin and psilocybin from mushrooms; and bufotenin and 5-MeO-DMT from toads.
The modified tobacco plants were found to produce all five compounds simultaneously. Because the different production pathways compete for the same resources, some compounds were produced in lower quantities than in their original sources.
However, the production was high enough to suggest that with a bit more tweaking, the system could function as a biological tryptamine factory for researchers.
Related: Single Dose of DMT Rapidly Reduces Symptoms of Major Depression
Berman, Höfer, and their team also took it a step further. By tweaking the enzymes involved in the tryptamine production pathway, the researchers were able to produce modified versions of the compounds that do not naturally occur in plants, and which may also have therapeutic value.
With further research, the system could be optimized to research requirements, or even help design new compounds tailored for specific therapeutic applications.
“Blending catalytic functions across the tree of life, coupled with metabolic engineering guided by rational protein design of mutant enzymes, enabled substantially more efficient in planta production of the indolethylamine components,” the researchers write.
“This work establishes a versatile platform for concurrent biosynthesis and diversification of psychoactive indolethylamines, paving the way for their production in plants.”
The research has been published in Science Advances.

