The University of Rochester Center for Health + Technology (CHeT) stands as one of academic medicine’s most successful experiments in translating science into patient benefit. Over nearly four decades, CHeT has played an instrumental role in bringing 12 drugs and devices to market—including landmark treatments for Parkinson’s, Huntington’s, and neuromuscular diseases—while pioneering new ways to design, measure, and deliver clinical trials.
What began as a small coordination effort in the late 1980s has evolved into a global model for how an academic research center can run industry-grade clinical trials, invent new endpoints, and move therapies from lab to life faster and more equitably.
Today, under the leadership of Chad Heatwole, MD, MS-CI, CHeT is building on that legacy while charting an ambitious path forward, employing new tools like digital endpoints and predictive analytics to speed clinical trials in neurological and other diseases. The approach blends scientific rigor with empathy for the patient experience. “Every new technology or trial design we adopt,” Heatwole said, “should be focused on improving the lives of the people we’re striving to help.”

At its core, CHeT embodies a simple but radical idea: that rigorous, patient-centered science and operational excellence can—and should—live within academia. From the analog coordination of early Parkinson’s trials to today’s decentralized, sensor-driven studies, CHeT’s evolution reflects a steady commitment to the principle that research should answer the questions that matter most to patients.
Building an academic powerhouse
CHeT’s origins at the University of Rochester date back to 1987, when neurologist Ira Shoulson, MD, launched the Coordination and Data Center to oversee DATATOP, an NIH study examining the effects of deprenyl (selegiline) and tocopherol (vitamin D) in Parkinson’s disease. The team enrolled roughly 800 participants across 28 sites, coordinating logistics by phone, fax, and mail—and learning early that patient welfare must drive design.

When the Data Safety Monitoring Board looked at the data and said it was unethical to not treat the people on a placebo, the study was restructured so all participants received active treatment. “We were developing research principles before it was mandated,” says Cindy Casaceli, MBA, who later guided the center’s operational maturation. Those experiences not only shaped the ethics of trial design but also laid the foundation for the sophisticated research enterprise that would follow.
Shoulson’s work also helped knit durable international networks of researchers—the Parkinson Study Group and the Huntington Study Group—that standardized practices, aggregated registries, and created recruitment pipelines still central to modern trials. Renamed the Clinical Trials Coordination Center (CTCC), as the 1990s progressed, the center professionalized, hiring industry talent to document workflows, building standard operating procedures, and demonstrating its ability to meet the industry’s regulatory expectations. Those changes transformed ad hoc academic competence into audit-ready operations, opening the door to partnerships with pharmaceutical firms.
Expansion, integration, and the emergence of CHET
By the 2000s, the team confronted a familiar problem: fragmented vendors, high indirect costs, and fragile supply chains that stalled trials. The response was pragmatic—build what was missing. In 2008, Cornelia Kamp, MBA, established the Clinical Materials Services Unit (CMSU) to provide cGMP-compliant packaging, labeling, distribution, and returns management. “We were getting really lousy service from big contract research organizations,” Kamp said. “So we built our own.” CMSU handled large-scale shipments, complex cold-chain logistics, and blinded-kit design, quickly attracting institutional and external business.

In 2009, Karl Kieburtz, MD, MPH, consolidated the CTCC, CMSU, and growing translational capabilities into the Center for Human Experimental Therapeutics (CHET)—a single entity to bridge “the valley of death” between discovery and first-in-human testing. Under Kieburtz, who had previously served as the first director of the CTCC, the entities moved out from under the Department of Neurology and into an independent center that brought these capabilities to the broader University and global academic community.
The unified center combined trial design, regulatory strategy, supply logistics, outcomes science, and analytics so investigators no longer had to stitch together vendors. With these capabilities in place, Rochester could now manage every phase of a trial—from design to delivery—under one roof. As Kieburtz put it, CHET offered industry “experience, expertise, and finesse.” That integrated model also became a training crucible, producing project managers, database programmers, and fellows who learned the full lifecycle of trials inside one system.
“Fifteen years of CHET/CHeT show how taking existing pieces of research infrastructure from within the University and positioning them in an independent center, along with new faculty and resources, can expand the mission and connectedness of research,” said Kieburtz.
The CHeT Era
Under the leadership of Ray Dorsey, MD, CHET evolved into CHeT—Center for Health + Technology—adding digital endpoints and decentralized methods to its toolkit while retaining the institutional engine that moves therapies toward patients.
Rebranding signaled more than a new name—it marked a shift from getting molecules into humans to reimagining how trials reach and measure patients. Under successive leaders—Kieburtz’s infrastructure build, Dorsey’s push into telemedicine and remote sensing, and now Heatwole’s emphasis on state-of-the-art measurement technologies and rare diseases—CHeT became a hybrid engine of science, operations, and technology.

CHeT’s portfolio still favors late-translational, multi-center studies—its “sweet spot”—but couples that work with digital tools and analytic muscle. CHeT associate director Jamie Adams, MD, and her team have driven trials that use wearable sensors, smartphone apps, and decentralized visits to capture continuous, real-world data. Charles Venuto, PharmD, founded and leads CHeT’s Analytics division, which allows researchers to model, predict, and act on data and design more efficient clinical trials. Brett Kinsler, DC, leads CHeT’s Health, a division that works to bring CHeT’s discoveries and technologies to the greater community, and Melissa Kostrzebski, MS, MBA, now heads the CTCC.
Operationally, CHeT is rare in scope: trial coordination, outcomes measurement, clinical materials, analytics, and regulatory support operate in concert, enabling the center to take on complex international protocols and novel endpoints that many partners consider too risky.
Defining the “CHeT Difference”
If CHeT’s brand can be distilled to one idea, it is this: measure what matters to patients. “Historically, treatment assessments have been created through black-room discussions in the absence of any patient input. As a result, the patient’s voice and preferences are often missing from research priorities,” said Heatwole.
Building on the NIH-supported National Registry for Myotonic Dystrophy & Facioscapulohumeral Dystrophy, in 2010, Heatwole created the first patient-reported outcome (PRO) measure for myotonic dystrophy, type 1. His team has since developed more than 250 validated, disease-specific PROs now employed by academic, industry, and government researchers around the globe to measure patient-meaningful responses to therapeutic interventions during clinical trials.
The second pillar is operational breadth. While some centers have fragments of CMSU’s capabilities, few have the whole package—from packaging and cold-chain logistics to interactive response systems and global distribution. CHeT can design sensitive endpoints, recruit hard-to-find patients, manage complex supplies, and stand behind data through regulatory review.
Transforming trial design: from clinics to connected devices
CHeT began integrating wearables, smartphone apps, and remote assessments early—because richer, continuous data and decentralized participation solve real, practical problems. Wearables capture continuous, objective signals over longer periods of time rather than snapshots in the clinic; they also expand access for participants who live far from academic centers. “By leveraging wearables, we’ve expanded the boundaries of clinical research,” said Adams, who is leading studies in Parkinson’s and Huntington’s that are employing smartwatches to measure progression in the diseases.
When paired with CHeT’s registries and modeling expertise, sensor data help identify likely responders, shrink sample sizes, and accelerate decisions. Regulators are watching. Though digital endpoints still require validation, CHeT’s approach—pairing patient-centered measures with continuous sensor streams—creates increasingly persuasive clinical trial dossiers. Connected devices have moved from experimental add-ons to core tools for modern trials.
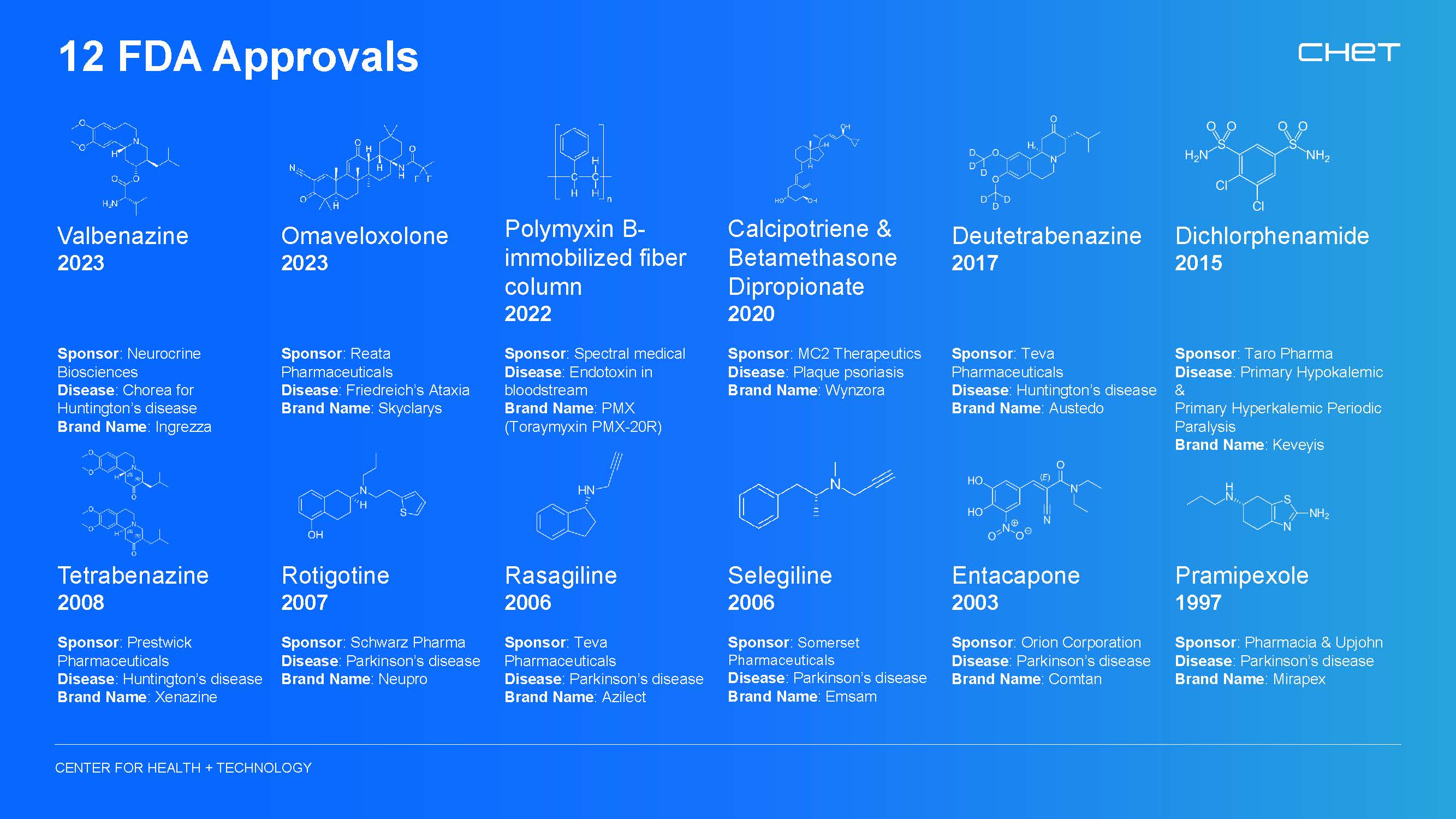
Real-world impact: approvals, rare diseases, and patients reached

The model is proven by treatments that reached patients and diseases once ignored by the pharmaceutical industry. Rochester has helped lead pivotal trials for five Parkinson’s drugs, three Huntington’s drugs, and treatments for rare neuromuscular diseases, among others. “Every drug approved to treat Huntington’s disease was studied through the University of Rochester,” Kieburtz said.
“While new therapies are still desperately needed, our researchers were involved in the development of most frontline drugs used to treat Parkinson’s, which have alleviated motor symptoms and improved the lives of people suffering from this progressive disease,” said Adams.
Beyond approvals, CHeT turned risky projects into viable programs by building natural-history registries, validating meaningful outcomes, and running inspection-ready operations. Casaceli captures the ethos: “We’re not just focused on finances…we’re focused on moving research forward and bringing hope to patients and families.”
As the center turns toward new frontiers—from brain-computer interfaces to AI-driven clinical trial modeling—it remains guided by the same principles that shaped its beginnings under Shoulson: scientific curiosity married to pragmatic compassion. That, ultimately, is the CHeT difference and the foundation for its next generation of innovation.
Originally published in NEUROSCIENCE Volume 27.
