What if the toughest part of wood could be turned into highly useful chemicals without burning fossil fuels or using high-pressure hydrogen? A new study sheds light on an interesting method to achieve this.
It proposes a clever electrochemical method to break down lignin—the rigid, stubborn material that gives plants their strength—into high-value compounds. This is a big deal because lignin is one of the most abundant carbon sources on Earth, yet it’s notoriously difficult to use.
“Its high aromaticity and strong chemical linkages render lignin highly recalcitrant, posing significant challenges for its efficient conversion,” the study authors note.
By replacing harsh conditions with electricity, this approach offers a simpler and cleaner way to unlock lignin’s potential. According to the researchers, if it works at scale, it could turn wood waste into a reliable source of fuels and valuable chemicals.
Rewiring lignin breakdown with electricity
Lignin has long resisted efficient conversion because of its tangled structure and very strong chemical bonds. Some of these bonds act like stubborn links holding the structure together, making them very hard to break.
Until now, breaking these links usually required high temperatures, high pressure, and added hydrogen gas. These harsh conditions consume large amounts of energy and still struggle to produce specific, useful products.
Earlier electrochemical approaches also faced limitations, including low yields and difficulty in clearly identifying the products formed. To overcome this, the researchers designed an electrochemical system built around a 5 wt% palladium-on-carbon (Pd/C) catalyst.
Instead of supplying hydrogen gas from outside, the system generates reactive hydrogen directly on the catalyst surface through an electricity-driven process involving water, enabling bond cleavage and hydrogenation to occur together.
This surface-bound hydrogen then attacks lignin’s tough bonds, breaking them apart while also transforming the fragments into more stable and valuable molecules. The team first tested this idea using model compounds that represent lignin’s key linkages.
Compounds mimicking 4–O–5 bonds, such as diphenyl ether and phenyl tolyl ether, were completely converted within 90 minutes at 70°C and a current density of 50 mA/cm².
“We achieved 100 percent conversion of diaryl ethers in 90 min with high selectivity of the corresponding monomers,” the study authors said. Even more impressively, another compound broke down fully at just 30°C, showing the method can work under milder conditions.
Moreover, the process didn’t just break the molecules—it also turned them into useful products with very high precision. In many cases, the desired chemicals were formed in more than 99 percent yield. These results show that once lignin’s tough bonds are broken, the system can efficiently turn the pieces into valuable chemicals.
A catalyst with two roles—and real biomass proof
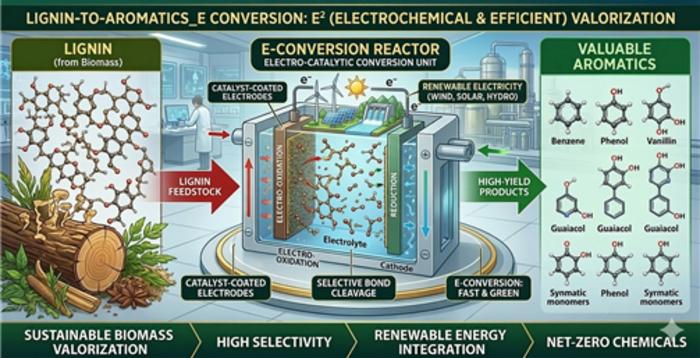
A key strength of this system lies in how the catalyst works. The Pd/C material operates through a dual-function mechanism. One form, palladium oxide (PdO), is responsible for breaking the carbon–oxygen bonds in lignin.
The other form, metallic palladium (Pd⁰), takes over afterward, hydrogenating the resulting fragments into stable compounds like cyclohexanol and cyclohexane. When tested separately, neither form performed well.
Pure palladium achieved only 19.3 percent conversion, while palladium oxide reached 57.4 percent. Together in Pd/C, they delivered the highest activity and selectivity.
The team then fine-tuned the process to make it work better. They found that adding a small amount of isopropanol (30 wt%) helped the reaction run more smoothly and use electricity more efficiently. The best results came at a current density of 50 mA/cm², while higher currents reduced performance by producing unwanted hydrogen gas.
To see if this works outside the lab, they tested it on real birch wood. After an initial step that removed 81 percent of lignin, only five percent of useful products were formed. However, when the new method was applied under milder conditions, the yield increased to 13.6 percent in one hour and 19.6 percent in four hours.
“We achieved 19.6 C% lignin-derived phenolic monomers yield from birch wood biomass,” the study authors added. Moreover, the process produced useful building-block chemicals (such as syringol and guaiacol derivatives) used in fuels and materials, with one key product making up 41.6 percent of the output.
A cleaner path to chemicals
This work introduces a new kind of biorefinery platform that runs on electricity instead of fossil fuels or high-pressure hydrogen.
By combining bond-breaking and chemical upgrading in a single step under relatively mild conditions, it offers a more sustainable way to turn plant waste into valuable materials. The ability to precisely control reactions using electrical input also makes the process flexible and potentially scalable.
However, there is room for improvement. The yields from real biomass, while improved, are still lower than what would be needed for large-scale industrial use.
Future research will likely focus on boosting efficiency and adapting the process for continuous, large-scale operation.
The study is published in the journal Applied Catalysis B: Environment and Energy.
