A familiar Alzheimer’s protein may have a hidden role in cell division.
Processes such as wound healing, hair growth, and the continual renewal of cells all rely on cell division. In this process, chromosomes must be evenly distributed between two daughter cells. Even small mistakes can lead to abnormalities within the cell.
Researchers at POSTECH (Pohang University of Science and Technology) have identified new evidence that a protein called tau may play a key role in maintaining this precision. Their findings were published in Nature Communications.
Tau is widely studied for its involvement in Alzheimer’s disease. In healthy neurons, it helps stabilize microtubules, which are thin structural fibers inside cells. In Alzheimer’s patients, however, tau can clump together abnormally. Recent studies have also shown that tau can gather molecules into small droplet-like structures known as condensates. Until now, its interaction with DNA has not been well understood, prompting the team to investigate further.
How Chromosomes Are Separated
During cell division, chromosomes are guided and separated by spindle microtubules, which pull them into two new cells. For this to happen correctly, chromosomes must attach accurately to these microtubules. To explore whether tau plays a role in this step, the researchers conducted experiments using individual DNA molecules.
Their results showed that tau binds to DNA and forms condensates. It can move along DNA strands and draw nearby strands together, suggesting a role in organizing chromosome structure.
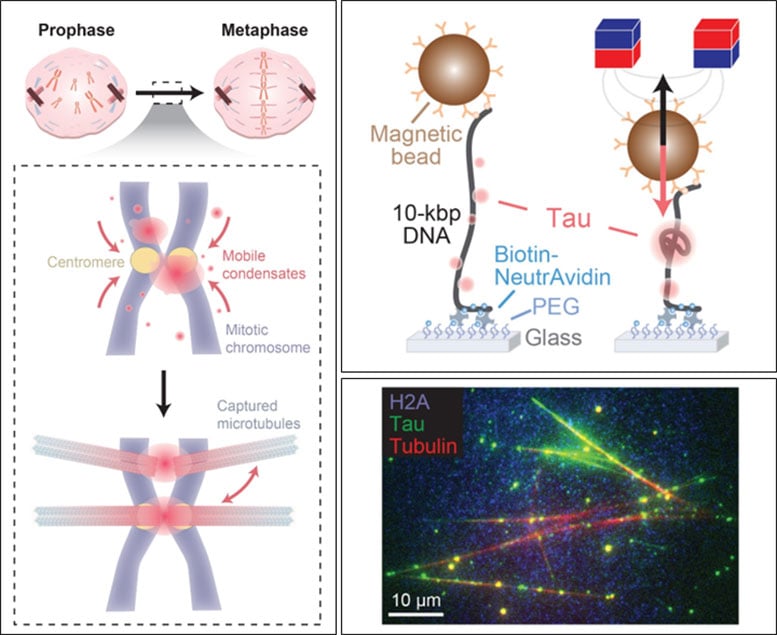
Using high-resolution fluorescence imaging, the team confirmed that these tau–DNA condensates act as connection points that can capture microtubules. This behavior was observed both in controlled laboratory conditions and in living cells.
The study also found that phosphorylation, a chemical change to tau, can influence this process. When cells produced a form of tau similar to that seen in Alzheimer’s disease, chromosomes often failed to align properly during division. This indicates that even minor changes in tau can disrupt the accuracy of cell division.
Broader Implications for Disease Research
The researchers believe these findings could open new directions for studying infertility, congenital conditions, and neurodegenerative diseases such as Alzheimer’s.
Professor Minju Shon, who led the study, said, “This work suggests that tau can directly interact not only with microtubules but also with DNA, potentially linking the two structures. It also shows that tau may be involved in the early stages when chromosomes first establish connections with spindle microtubules during cell division.”
Reference: “Tau condensation on DNA mediates microtubule attachment suggesting a mitotic role for centromere-localized tau” by Celine Park, Jaehun Jung, Yuri Hong, Haeun Yoo, Keunsang Yang, Jaehyeon Shin, Minsik Kim, Chan Lim, Ayoung Jeong, Seokyun Hong, Jun Young Baek, Sang-Hyun Rah, Chaelin Lee-Eom, Minseok Seo, Yoori Kim, Jae-Hyung Jeon, Jong-Bong Lee, Dong Soo Hwang and Min Ju Shon, 7 January 2026, Nature Communications.
DOI: 10.1038/s41467-025-67888-x
This research was supported by the Basic Science Research Program of the National Research Foundation of Korea (NRF), funded by the Ministry of Science and ICT (Outstanding Young Researcher Program and Basic Research Laboratory Program).
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.
