By watching Alzheimer’s-related protein clumping unfold second by second, researchers have uncovered new clues about the role of metal ions.
An Oregon State University chemist and a group of undergraduate researchers have captured real-time details of a chemical reaction tied to Alzheimer’s disease. Their work could help guide the development of more precisely targeted medications.
In laboratory experiments, the team used a molecular measurement method to track how certain metals encourage amyloid-beta proteins to clump together. These clumps are associated with the breakdown of neural communication seen in Alzheimer’s patients.
The study was led by Marilyn Rampersad Mackiewicz, an associate professor of chemistry in the OSU College of Science. In addition to monitoring how metals trigger protein buildup, the researchers examined how compounds called chelators can interfere with or even undo that process.
The findings were published in ACS Omega.
Understanding Alzheimer’s and Protein Aggregation
Alzheimer’s disease is the most common type of dementia. It is a long-term condition that gradually damages memory and thinking skills, affecting millions of older adults and their families. The Centers for Disease Control and Prevention reports that Alzheimer’s is the sixth-leading cause of death among people age 65 and older.
A hallmark of the disease is the accumulation of amyloid-beta proteins in the brain. When these proteins cluster together, they disrupt communication between nerve cells. Although metals such as copper are essential for normal brain function, trouble can arise when their levels become unbalanced.
“Too many of some metal ions, like copper, can interact with amyloid-beta proteins in ways that lead to protein aggregation, but most experiments have only shown the end result, not the interactions and aggregation process itself,” Mackiewicz said. “We developed a method that lets us observe those interactions live, second by second, and directly measure how different molecules interrupt or reverse them. It shifts the question from ‘does something work?’ to ‘how does it work, and when?’”
The Role of Chelators
Chelators are molecules that bind tightly to metal ions. The term comes from the Greek word for claw, reflecting how these compounds grasp metals at the molecular level.
Using a technique called fluorescence anisotropy, the scientists evaluated two different chelators. One successfully captured metal ions but did so broadly, without distinguishing between metals that contribute to amyloid-beta aggregation and those that do not.
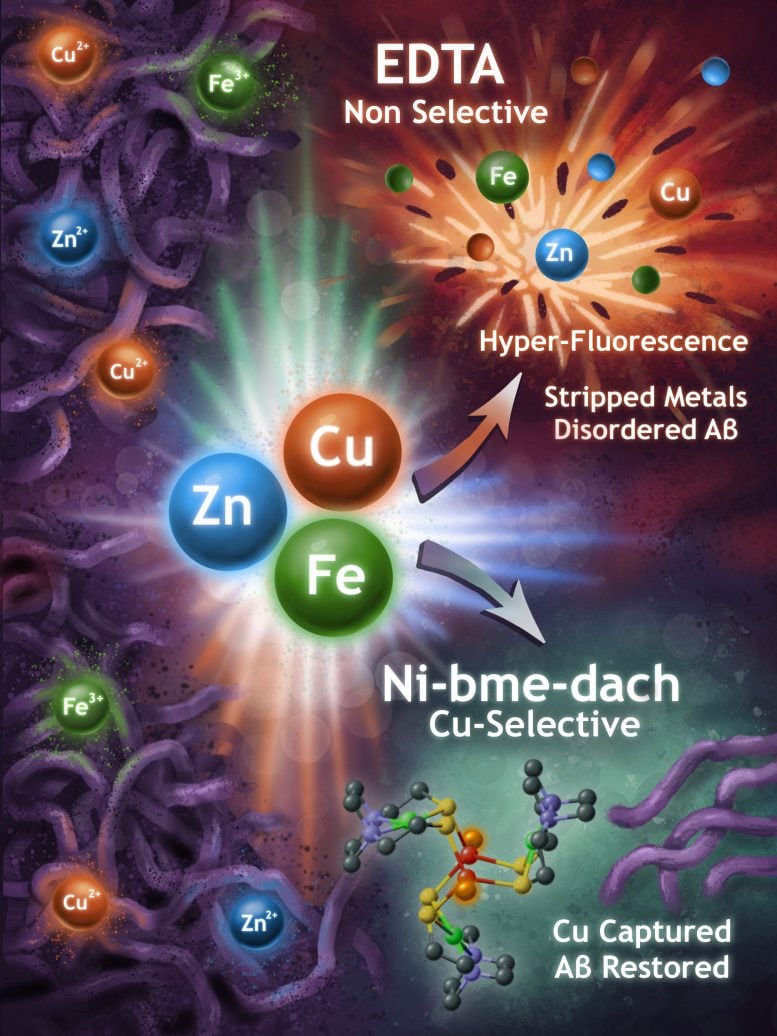
The second chelator behaved differently. It showed a strong preference for binding copper ions, which are thought to play a key role in Alzheimer’s-related protein buildup.
Implications for Treatment Development
“That kind of real-time insight into how the protein aggregations form and unform is important for designing better treatments and for understanding why some widely used chemical approaches may not behave the way we assume they do,” Mackiewicz said. “Alzheimer’s affects millions of families and while clinical treatments based on this work remain years away, discoveries like this can offer genuine hope – with the correct targeting, some of the brain damage might be reversible.”
According to Mackiewicz, the next phase of the research will involve testing the findings in more complex biological systems, including cellular and preclinical models.
“Many potential Alzheimer’s treatments fail due to an incomplete understanding of how amyloid-beta protein aggregation occurs,” she said. “By directly observing and quantifying these interactions, our work provides a roadmap for creating more effective therapies.”
Reference: “Selective Reversal of Cu-Amyloid Aggregation Monitored in Real Time by Fluorescence Anisotropy: Ni-Bme-Dach vs EDTA Benchmarks” by Alyssa N. Schroeder, Eleanor K. Adams, Dane C. Frost, Erica Lopez, Jennie R. Giacomini and Marilyn R. Mackiewicz, 7 February 2026, ACS Omega.
DOI: 10.1021/acsomega.5c11345
Support from the SURE Science Program and donors Julie and William Reiersgaard made possible the research contributions of undergraduate students Alyssa Schroeder of OSU and Eleanor Adams, Dane Frost, Erica Lopez and Jennie Giacomini of Portland State University.
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.
