A hidden effect of statins on muscle cells may explain why some patients cannot tolerate them.
For millions of people, statins are a daily safeguard against heart attacks and strokes. But for about 1 in 10 users, these widely prescribed drugs come with an unexpected tradeoff—persistent muscle pain, fatigue, and weakness that can be severe enough to make patients stop taking them altogether.
New research from scientists at Columbia University and the University of Rochester is shedding light on why this happens. Their findings point to a breakdown in how muscle cells handle calcium, offering a clearer explanation for statin-associated muscle symptoms (SAMS) and suggesting new ways to prevent them.
Statins lower cholesterol by blocking a liver enzyme needed to produce it, which reduces levels of LDL, often called “bad” cholesterol. This helps slow or prevent atherosclerosis, a condition in which fatty deposits build up inside blood vessels and restrict blood flow. Despite these benefits, statins do not act exclusively on the liver. They also interact with other proteins throughout the body, including one that plays a central role in muscle function.
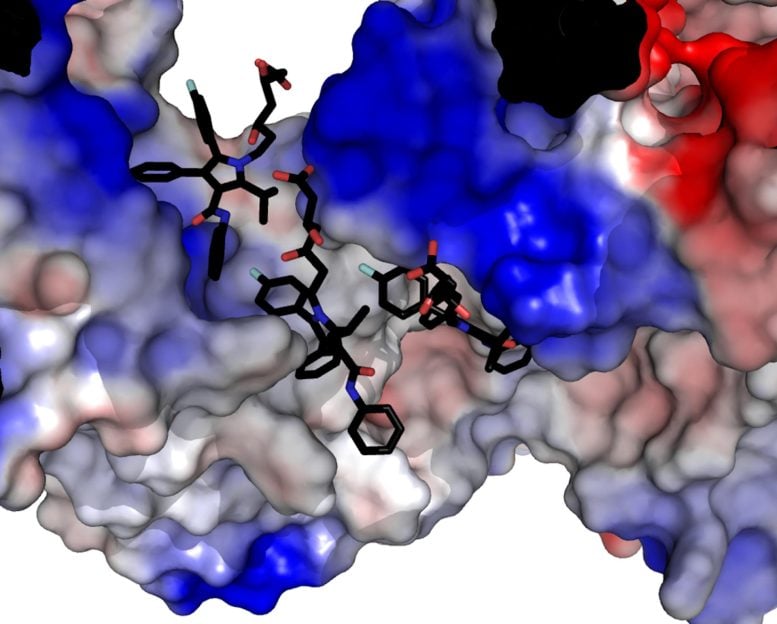
That protein, known as ryanodine receptor 1 (RyR1), sits within the sarcoplasmic reticulum, a network inside muscle cells that stores calcium. Under normal conditions, RyR1 tightly controls when calcium is released, allowing muscles to contract and relax in a coordinated way. Even small disruptions to this system can have noticeable effects, since calcium signaling is fundamental to nearly every muscle movement, from walking to breathing.
Visualizing the Molecular Interaction
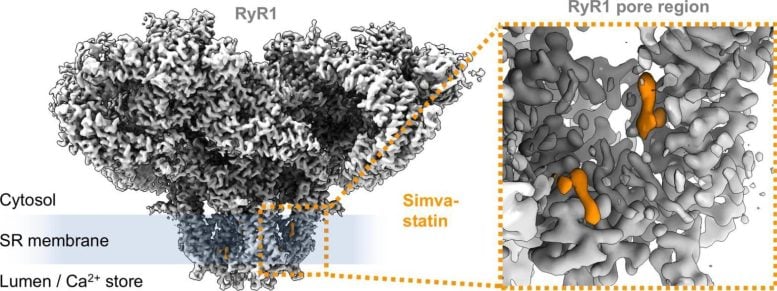
Using mice as a model and high-resolution cryo-electron microscopy, the researchers captured how statins physically interact with this channel. The imaging method involves rapidly freezing biological samples and analyzing them with electron beams to reconstruct detailed three-dimensional structures at the molecular level.
The team found that certain statins, including simvastatin, can bind to RyR1 in a way that destabilizes the channel. Instead of opening only when needed, the channel becomes “leaky,” allowing calcium to seep continuously into muscle cells. This excess calcium can directly damage muscle fibers or activate enzymes that begin breaking them down.
The result is a range of symptoms familiar to many patients, including soreness, cramping, tenderness, and reduced strength. In more vulnerable individuals, especially those with genetic mutations affecting RyR1, the consequences can be more serious. These patients may face complications such as malignant hyperthermia, a dangerous spike in body temperature triggered by certain drugs, or impaired diaphragm function that affects breathing.
In rare cases, the damage escalates into rhabdomyolysis, a condition in which muscle tissue breaks apart and releases its contents into the bloodstream, potentially leading to kidney failure. Another uncommon but severe reaction is autoimmune-mediated necrotizing myositis, where the immune system mistakenly attacks muscle tissue.
A Widespread Issue With Clinical Impact
This calcium leak mechanism may not explain every instance of statin intolerance, but it provides a major piece of the puzzle. With roughly 40 million adults in the United States taking statins, even a partial explanation could have wide-reaching impact.
“I’ve had patients who’ve been prescribed statins, and they refused to take them because of the side effects,” says lead author Andrew Marks, a cardiologist at the Columbia University Vagelos College of Physicians and Surgeons. “It’s the most common reason patients quit statins, and it’s a very real problem that needs a solution.”
The findings open up two promising paths forward. One approach is to redesign statins so they continue to block cholesterol production without interfering with RyR1.
Another option is to protect the muscle cells directly. In the study, researchers tested an experimental class of drugs called Rycals in mice that could not tolerate statins. These compounds helped stabilize the calcium channel, preventing leaks and reducing muscle weakness.
Reference: “Cryo-electron microscopy reveals sequential binding and activation of Ryanodine Receptors by statin triplets” by Steven Molinarolo, Carmen R. Valdivia, Héctor H. Valdivia and Filip Van Petegem, 20 November 2025, Nature Communications.
DOI: 10.1038/s41467-025-66522-0
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.
